No doubt an election result lent some strength to yesterday’s record high market open, but I’d say the exciting news that Pfizer Inc. (NYSE: PFE) and BioNTech SE ADR’s (NASDAQ: BNTX) joint novel coronavirus vaccine candidate appears to be 90% effective will have even more impact, though the true nature of that impact isn’t quite as straightforward as Big Media is telling us.
I couldn’t be happier to hear the news about Pfizer and BioNTech’s interim data, but we have to be realistic: Neither the “vaccine race” nor the pandemic are over by any stretch of the imagination.
Unfortunately, some investors have drawn the wrong conclusions – or perhaps “leapt” to the wrong conclusions is the better word.
US Global Jets ETF (NYSEArca: JETS), which I warned readers away from just last week, jumped nearly 23% at market open; one of the exchange-traded fund’s biggest holdings, American Airlines Group Inc. (NASDAQ: AAL) gapped up more than 25% at the opening bell.
Investors seem to have reasoned that the travel sector will come roaring back when we get a vaccine, and ultimately they’re right – but right now they’re dangerously early, too.
Like the vaccine race itself, recovery for the travel sector and indeed the entire economy will be a marathon, not a sprint.
While my recommendation to sell JETS and other airline and travel stocks still stands, I think every investor needs to understand the real state and stakes of the vaccine race – where we’re going, when we might get there, and how best to get positioned for profits…
There’s a Light at the End of the COVID-19 Tunnel
“It is not the end. It is not the beginning of the end. But it is, perhaps, the end of the beginning.” Britain’s great wartime Prime Minister, Winston Churchill, uttered that famous quote after the Battle of El Alamein – the very first big, important British and Commonwealth victory in North Africa.
It was a huge morale boost, naturally. Folks were excited to see the good guys winning – for a change – but Churchill of course wisely knew that there were years and years of hard, bitter fighting left to do.
And that perfectly sums up where yesterday’s Pfizer news puts us in the race for the coronavirus vaccine which, not unlike World War II, is a mammoth multinational undertaking involving a significant percentage of the planet’s scientific skill and talent.
In other words, the news of Pfizer’s mRNA candidate vaccine is a positive, inspirational development – particularly as we see cases soar at home and abroad – but there are long, tough months ahead.
Exuberant investors who invest as if it’s actually “the beginning of the end,” will face a tough reckoning.
To start, there’s the data itself. Government health officials were prepared to approve a vaccine with effectiveness as low as 50%, but that 90% figure is entirely preliminary; by the time the safety and efficacy data are submitted, that number could be lower. Of course, it could be even higher. We just don’t know. We don’t know how long immunity lasts, neither do we know whether the folks who do catch coronavirus – there are always some with every vaccine – after vaccination have a less serious disease.
And there is a massive logistical challenge ahead, too. The Pfizer/BioNTech candidate requires two shots a few weeks apart, and those shots must be kept in the “cold chain” of storage. By all accounts, the Pfizer/BioNTech candidate needs to be stored at -94 degrees Fahrenheit (you read that right: ninety-four degrees below zero) from the time it leaves the manufacturer to the time it ends up in a human body. Pfizer and BioNTech are looking into whether there’s any leeway there, but few if any countries have that capability on the scale required to vaccinate billions of people.
The good news is, all over the world, organizations and governments are taking heroic strides to get us this kind of capability. Here at home, the U.S. military – the sharpest, most talented logistics outfit this side of FedEx – is tackling the problem as part of its role in “Operation Warp Speed.”
Healthcare workers are almost certainly going to be prioritized. According to the latest government data, some 18 million people work the U.S. healthcare sector alone, and we’re likely to have just 50 million doses on hand by the end of 2020, or enough for 25 million people. Pfizer has agreed to supply Uncle Sam with 100 million doses to start, with an option for 500 million more, which would cover most of the country’s population.
Pfizer has inked deals with the European Union to provide 200 million doses, with an option for 100 million more, which would cover the majority of the union’s 446 million citizens. With Japan, Pfizer has agreed to supply 120 million doses, which would, again, cover the majority and lend support to “herd immunity.”
All told, we should have access to 1.3 billion doses, or shots for 650 million people, by the end of 2021.
That late 2021 target date correlates to the growing consensus among health officials that we’ll be able to reclaim at least some of our normal lives by the third quarter of 2021.
That will be the real “beginning of the end” when it comes to the devastating global pandemic that’s claimed 1.26 million lives; around 238,000 of those have died in the United States.
Pfizer and BioNTech are most definitely in a favorable position here, but when it comes to these vaccines, more is definitely better. They’re not alone…
Pfizer and BioNTech Are in Very Good Company
Moderna Inc. (NASDAQ: MRNA) is also working on the same kind of revolutionary, never-before-seen mRNA vaccine – the method uses messenger RNA (hence the ticker) to turn your own cells into “vaccine factories” – as Pfizer and BioNTech. Their success makes it that much more likely Moderna’s candidate will also be successful. If true, it would be Moderna’s first approved drug – astonishing when you consider how popular this stock was with the mobile investing crowd over the summer. Moderna’s vaccine would also follow the two-dose/one-person rubric.
We could see the same kind of phase 3 data from Moderna within the next three weeks. The company says it’s ready to ship doses for 10 million people by the end of 2020 and is currently building capacity to manufacture 1 billion doses by the end of 2021. If Moderna’s candidate is proven safe and effective, the government has agreed to spend $1.2 billion to buy doses for 50 million people. The firm has secured commitments to supply the governments of Japan, Canada, Switzerland, Israel, and Qatar, too.
There’s the “Boston vaccine,” too, under development by Johnson & Johnson (NYSE: JNJ) and Beth Israel Deaconess Medical Center. Interestingly, this group has the only phase 3 candidate that only requires one dose for effectiveness; that’s potentially a massive production and distribution advantage. The vaccine is already effective in monkeys, but human results are expected by the end of 2020. The group is building manufacturing capacity of at least 1 billion doses by the end of 2021, with deals to supply the United States with 100 billion doses and the EU with 200 million.
AstraZeneca Plc. (NASDAQ: AZN) and Oxford University have shown their vaccine is effective in monkeys, and phase 3 results on humans are due by the end of December. The consortium has entered into a production deal with India’s Serum Institute, where production of millions of doses is already underway – and has been since the results of the primate trials.
That rapid production buildout isn’t quite “buying sight unseen,” but it is a leap of faith – one that may pay big dividends in the race for production. AstraZeneca/Oxford’s production target is 2 billion doses per year, of which 400 million has been earmarked for the EU.
And rounding out the front of the pack is Maryland-based NovaVax Inc. (NASDAQ: NVAX), whose two-dose/one-person candidate has produced “promising” results in monkey and human trials. NovaVax’s candidate uses a technology that was successful in producing its influenza vaccine last March.
Of all the leaders in the field, NovaVax has the longest distance yet to run; its UK phase 3 trial should see results in early 2021, and there’s a much larger U.S. phase 3 study starting this month. It’s inked deals with the Australian government to cover 40 million doses and 100 million doses for the U.S. government by the end of Q1/2021. NovaVax has also wisely secured the assistance of India’s Serum Institute to boost production.
And so investors have a few tradeoffs and advantages to consider. Johnson & Johnson, for instance, has the edge with its fewer required doses, whereas AstraZeneca and NovaVax have higher production capacity. Pfizer, BioNTech, and Moderna are further along.
Here’s How to Play It
Of the frontrunners, we can expect Pfizer/BioNTech, Moderna, AstraZeneca/Oxford, and Johnson & Johnson to return data and file for the all-important Emergency Use Authorization from the U.S. Food & Drug Administration by the end of this year.
Together, they have the capacity, or will have the capacity, to make enough vaccines for around 2.15 billion people, assuming things run smoothly – as, frankly, they have so far. That means we could have around 300 million Americans immunized against coronavirus in 2021, which would take us a long, long way toward returning to “normal.”
That said, BioNTech, a stock I first recommended in late summer, is the smartest buy here. I can also recommend Moderna for the simple fact that it’s using the same revolutionary technology.
As massively disruptive as the pandemic is, mRNA vaccines will have implications far, far beyond the novel coronavirus in treating all kinds of viruses.
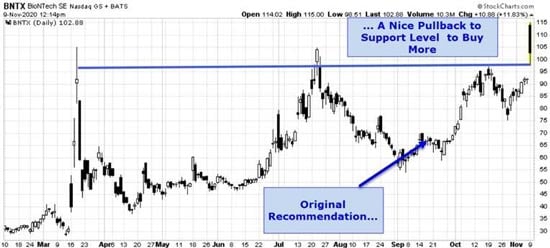
The technical picture on BNTX is extremely favorable right now; there’s a beautiful entry point available. If you move now, you’ll only enjoy bigger and bigger profits as the vaccine race moves into a new, decisive phase.
— D. R. Barton, Jr.
Source: Money Morning


